how to draw blood sample from dog
Sampling Overview


Do Not Submit:
EDTA or heparin plasma, serum, serum separator or clot activator tubes.
These samples are invalid for coagulation screening tests, protein C, or gene assays.
Vacutainer Describe - Large Dogs
Draw blood with a vacutainer needle or butterfly catheter directly into a 3.ii% or three.8% citrate tube (blue summit tube) Make sure the tube is in-engagement and completely filled past vacuum describe.
Syringe Draw - Small Dogs/Cats
- Draw an exact volume of citrate into a syringe to obtain 1 of the following full samples:
0.ii ml citrate + 1.8 ml blood = 2.0 ml total
0.3 ml citrate + ii.seven ml blood = 3.0 ml total
0.4 ml citrate + 3.six ml claret = 4.0 ml total - Perform venipuncture to collect full sample book.
- Transfer citrated blood sample to a plastic or no-additive drinking glass tube for centrifugation.

Plasma Separation
- Centrifuge the whole claret sample for 10 to 15 minutes.
- Aspirate plasma and transfer to a plastic or no-condiment drinking glass shipping tube.
If clot fragments or hemolysis (unless in vivo hemolysis) –redraw - Refrigerate plasma for same-day shipment or store frozen for upwardly to 2 weeks.

Shipping
- Send for overnight delivery on frozen common cold packs.
- Place the common cold pack and sample tube in a plastic bag
Afix tube to cold pack with condom band, wrap advisedly to prevent breakage - Fill all dead space with crumpled paper or bubble wrap
- Consummate an AHDC submission form, place in the outer box
Sampling Instructions
Collection Method 1: Vacutainer method
Materials: 3.0 ml sodium citrate (blue top) Vacutainer tube, Vacutainer needle or Vacutainer butterfly (22 or 21gauge), dilute nolvasan or antiseptic wipe,
Options: either iii.eight% or 3.ii% sodium citrate tubes can be used, larger volume (4 to 5 ml describe) tubes tin be used for big (>50 lb.) dogs, hair overlying venipuncture site tin can be clipped
Procedure:
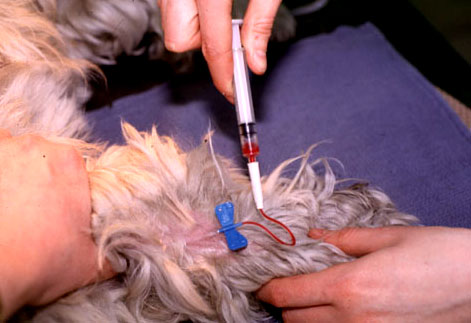
- Gently restrain the patient and apply manual pressure proximal to the cephalic or saphenous venipuncture site to permit the vein to fill with blood.
- Perform venipuncture using the Vacutainer needle or butterfly
- Attach the tube and allow blood to fill up the tube completely by vacuum
- Withdraw the needle and utilise direct pressure to the venipuncture site
Drove Method ii: Syringe method
Materials: sodium citrate anticoagulant, three.0 ml syringe, 22 or 21 approximate needle or 22 or 21 gauge brusk tubing butterfly catheter (short tubing = 3.5 inches), dilute nolvasan solution or antiseptic wipe
Options: either iii.8% or 3.2% sodium citrate tin exist used, larger volume (half-dozen ml syringe could be used to collect 4 to 6 ml sample volume) hair overlying venipuncture site can exist clipped
Procedure:
- Gently restrain the patient and utilise manual pressure proximal to cephalic or saphenous venipuncture site to allow the vein to fill with claret.
- Aspirate an exact book of citrate into the collection syringe to give a concluding ratio of 1 role citrate to 9 parts blood. Use one of the following sampling protocols:
2.0 ml full sample book = 0.2 ml citrate + 1.8 ml blood
iii.0 ml total sample volume = 0.3 ml citrate + 2.vii ml blood
4.0 ml total sample volume = 0.4 ml citrate + 3.6 ml claret - Perform venipuncture and collect blood into the syringe containing pre-measured citrate to obtain the exact total sample book.
- Withdraw the needle and use direct pressure level to the venipuncture site
- Remove the needle from the drove syringe and gently express the blood sample into a plastic tube.
Notes on Collection Methods
- Use of peripheral veins, rather than the jugular vein, is preferred for patients having severe hemostatic defects because of the risk of hematoma formation at venipuncture sites. Cervical hematoma formation could compromise the patient's airway.
- Claret samples can be drawn from an indwelling catheter using the syringe method, if the catheter is first flushed with 5 to ten ml. sterile saline (0.9% saline, NO HEPARIN). A 3 to 5 ml aliquot of claret should exist withdrawn from the catheter, and so the sample syringe containing pre-measured citrate is attached to the catheter and blood is withdrawn to obtain the exact total sample volume.
- Atraumatic venipuncture technique is critical to prevent activation/depletion of clotting factors during collection.
- The ratio of citrate to blood is critical for valid results. The sample should exist redrawn if the Vacutainer tube or syringe is not filled to the desired sample book.
- Sodium citrate (3.2 or three.8%) is the only acceptable anticoagulant for coagulation assays. Samples drawn into EDTA, heparin, or other anticoagulants or samples fatigued into clot activator or serum separator tubes are invalid for coagulation testing.
Processing Instructions for Coagulation Assays
Materials: Tabletop centrifuge, plastic pipettes or TB syringes, plastic tubes with secure caps, waterproof marker for labeling.
Process:
- Centrifuge the citrate whole claret sample tube for ten to 15 minutes at loftier speed (>2,500 g).
- Aspirate the supernatant plasma with a plastic pipette or TB syringe and transfer the plasma to a clean empty plastic tube.
- Label the sample tube with patient identifiers.
- Transport the sample to the testing laboratory inside one/2 hour of processing or shop the sample frozen until ship or shipment to the testing laboratory.
Notes on Processing Methods
- Siliconized glass tubes tin be used as centrifugation and storage tubes, however glass tubes may crack, or shatter.
- The presence of clots or jell fragments in the sample is an indication of improper claret drove. Hemolyzed plasma is an indication of improper sampling, unless the patient has a hemolytic disease process. Samples should exist redrawn for valid results.
Source: https://www.vet.cornell.edu/animal-health-diagnostic-center/laboratories/comparative-coagulation/sampling-instructions
Posted by: formanthoulace.blogspot.com


0 Response to "how to draw blood sample from dog"
Post a Comment